When pathogens (such as bacteria, viruses, etc.) invade the body, B cells secrete antibodies (also known as immunoglobular proteins) to neutralize the antigen. An antibody protein is a Y-shaped molecule consisting of four polypeptide chains (two heavy chains and two light chains). The Y-shaped structure contains an antibody binding site (paratope, which can be interpreted as a “lock”) at each of the two tips. Each antibody binding site can specifically bind an antigen-epitope (which can be understood as a “key” to a “lock”)[1]. The unique and specific correspondence between antibody-binding epitope and antigen-epitope is the basis of the high specificity of antibody-antigen binding. Antibodies are widely used in pharmaceutical research and development. There are two main classes of antibodies: monoclonal antibody (Mabs) and polyclonal antibody (Pabs)[2].
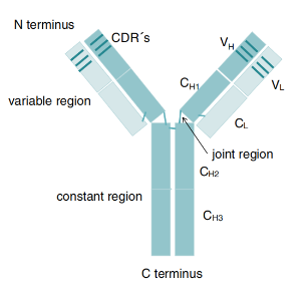
Figure 1: Antibody structure
Polyclonal antibodies are mixtures of antibodies produced by different B cells in the body against the same antigen. They can recognize and bind different epitopes on that single antigen. Polyclonal antibodies are produced by injecting certain immunogens into animals. After animal immunization, polyclonal antibodies can be obtained directly from serum (blood after removal of coagulation proteins and red blood cells) or further purified to remove other serum proteins. The specific process includes preparing antigens, immunizing animals, collecting serum after qualified titer determination, purifying and identifying antibodies. The process is simple, low cost and fast[3].

Figure 2: Method of production of polyclonal antibodies
Monoclonal antibodies are produced by identical B cells (clones of the same mother cell). This means that monoclonal antibodies can only recognize a specific single epitope. Unlike polyclonal antibodies, which are produced directly by animals, monoclonal antibodies are produced using tissue culture techniques in vitro. The process is as follows: ① Animal immunity and PBMC isolation. Cows are selected as experimental animals and are immunized 4-5 times by injecting a target antigen (usually a target protein or polypeptide) to stimulate an immune response, thereby activating the cow’s immune system and causing B cells to produce antibodies. Then ELISA assay, peripheral blood collection and PBMC isolation were performed. ② Flow sorting. Antigen specific single B cells were sorted by PBMC flow sorting method. ③ Single B cell culture screening. The single B cells were first cultured in 5-20 plates with 96-well plates in vitro, and then the supernatant of the cell culture was screened by ELISA or pre-interventional detection such as FACS. ④ Sequencing of positive clones. At least 20 positive cell clones were selected for RT-PCR to amplify their antibody variable region genes and then sequenced. ⑤ Vector construction, antibody expression and purification. Finally, the mammalian expression vector, cell transfection and ELISA were verification.
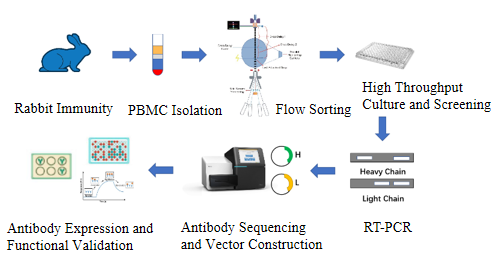
Figure 3: Method of production of monoclonal antibodies
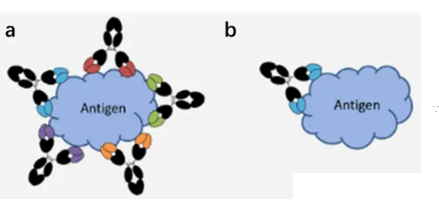
Figure 4: The mode of binding of (a) polyclonal antibody to (b) monoclonal antibody to antigen
Monoclonal antibody and polyclonal antibody have their own distinct characteristics and advantages. The specificity of monoclonal antibody is high, and once successfully prepared, both permanent monoclonal antibody and multi-clonal antibody have their own distinct characteristics and advantages. Monoclonal antibody has a high specificity, and once it is successfully prepared, it can sustainably produce completely consistent monoclonal antibody, so its specificity can be comprehensively and systematically verified. However, if the identified epitope is destroyed, the results of the experiment will be greatly affected, which is also one of the disadvantages of monoclonal antibodies. However, the specificity of polyclonal antibodies is poor, even if the same antigen is used to prepare polyclonal antibodies, there will be differences between different batches, so there are great limitations in specificity and consistency. Therefore, when using multiple antibodies for immune detection, it is easier to create a background, such as a mixed band in WB, a darker background in IHC, and so on. Although there is still a crossover. However, because multiple antibodies recognize multiple epitopes, even if a few epitopes are destroyed or the antigen conformation is changed, the results of the experiment will not be affected. Under the same conditions, the use of multiple antibodies can improve the sensitivity of the detection, and it is easier to detect proteins with low abundance.
References
[1] Lipman NS, Jackson LR, Trudel LJ, Weis-Garcia F. Monoclonal versus polyclonal antibodies: distinguishing characteristics, applications, and information resources. ILAR Journal. 2005;46(3):258-268. doi:10.1093/ilar.46.3.258
[2] Bregenholt S, Jensen A, Lantto L, Hyldig S, Haurum JS. Recombinant human polyclonal antibodies: a new class of therapeutic antibodies against viral infections. Curr. Pharm. Des. 12(16), 2007–2015 (2006). doi: 10.2174/138161206777442173
[3] Grodzki AC, Berenstein E. Antibody purification: ammonium sulfate fractionation or gel filtration. Methods MolBiol. 2010;588:15-26. doi:10.1007/978-1-59745-324-0_3
